Plant hormones move silently through tissues, yet they control almost every aspect of plant life, from root growth and leaf expansion to flowering, stress responses, and survival under extreme environments. For decades, plant physiologists knew that hormones such as auxin, abscisic acid, cytokinins, gibberellins, ethylene, and jasmonates must travel from one cell to another to exert their effects. However, directly observing this movement inside living plant cells remained one of the greatest challenges in plant biology. Traditional approaches relied on indirect measurements: hormone extractions, reporter gene expression, mutant analysis, or radioactive labeling. While these methods provided valuable insights, they lacked spatial and temporal precision. Hormone transport is dynamic, highly localized, and often changes within seconds or minutes. Capturing these rapid events required a new generation of tools.
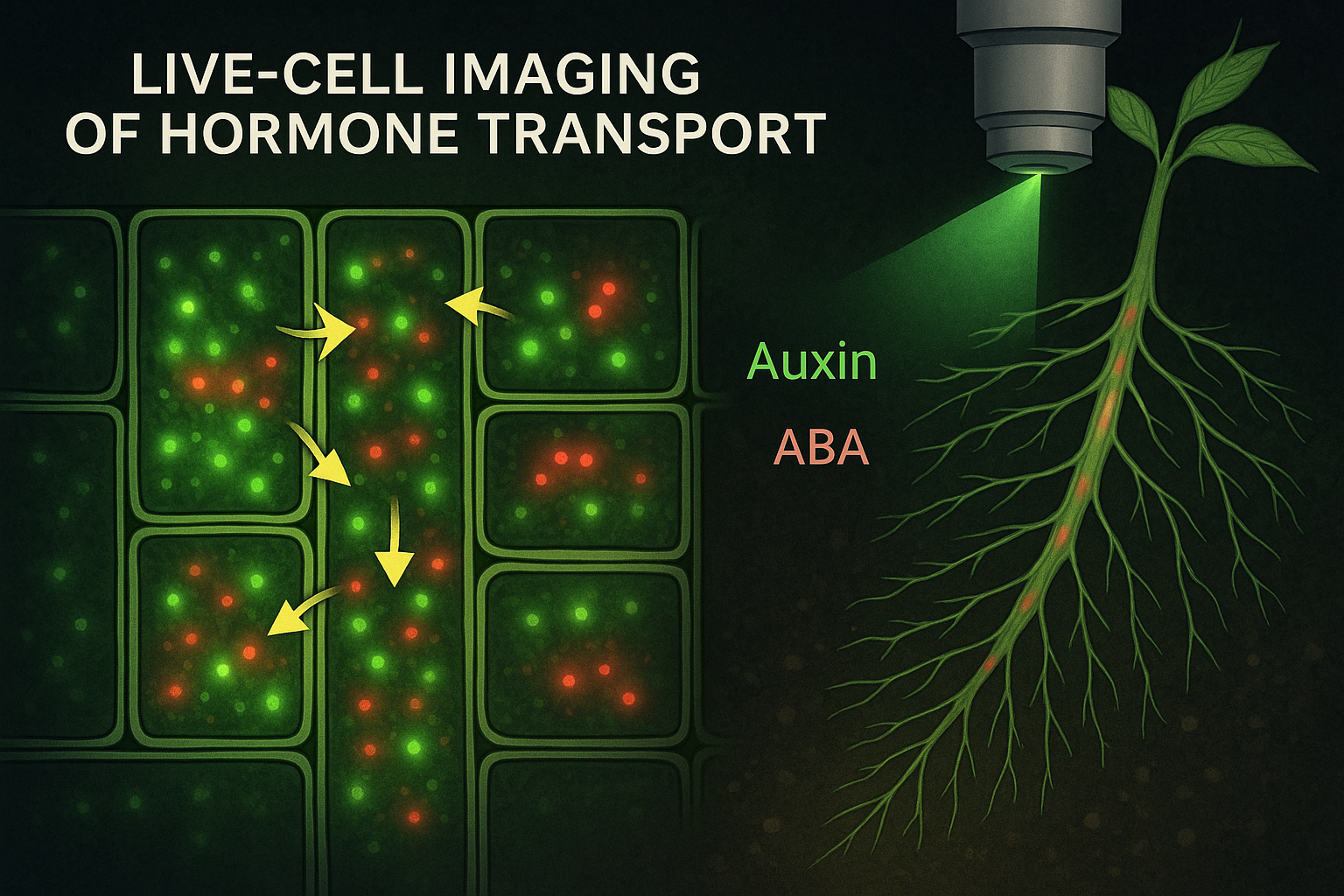
Live-cell imaging of hormone transport has transformed this field. By combining fluorescent reporters, biosensors, advanced microscopy, and genetic tools, researchers can now visualize hormone movement in real time inside living cells and tissues. This technology has revealed that hormone transport is far more dynamic, regulated, and context-dependent than previously imagined. It has reshaped our understanding of plant development, stress physiology, and long-distance signaling.
Why Hormone Transport Matters in Plant Physiology
Hormones are not evenly distributed throughout a plant. Their effects depend on where they are synthesized, how they move, where they accumulate, and how cells interpret their signals. Auxin gradients guide root and shoot architecture. Abscisic acid redistributes rapidly during drought to close stomata. Cytokinins move from roots to shoots to regulate growth and senescence. Jasmonates and salicylic acid spread following wounding or pathogen attack to activate defense responses. Hormone transport is therefore not just a delivery system; it is a regulatory mechanism. The direction, speed, and localization of hormone movement determine developmental patterns and stress responses. Small changes in transport can lead to dramatic phenotypic effects.
Live-cell imaging has shown that hormone transport is not a simple diffusion process. It involves specific transporters, vesicle trafficking, cell polarity, cytoskeletal dynamics, and feedback regulation. Transport proteins themselves move, recycle, and change localization in response to environmental cues. Without live imaging, these dynamic behaviors would remain invisible. Understanding hormone transport at the cellular level is especially important in fluctuating environments. Under drought, heat, or salinity, plants must rapidly redirect hormonal signals to balance growth and survival. Live-cell imaging allows researchers to see how these signals move in real time, offering insights that static measurements cannot provide.
Tools and Technologies Behind Live-Cell Hormone Imaging
The ability to visualize hormone transport relies on a powerful combination of molecular reporters and advanced microscopy. One of the most important breakthroughs was the development of fluorescent hormone reporters. These reporters use fluorescent proteins to indicate hormone presence or signaling activity within specific cells. Auxin research led the way. Reporters such as DR5 and later degradation-based sensors made it possible to visualize auxin response patterns with cellular resolution. More recently, direct auxin sensors have allowed researchers to detect changes in auxin concentration rather than just downstream signaling. These sensors reveal rapid auxin redistribution during tropic responses, organ initiation, and stress. For abscisic acid, genetically encoded biosensors have enabled real-time visualization of ABA accumulation in guard cells, vascular tissues, and roots. These sensors have revealed how quickly ABA levels rise during drought and how spatial patterns of ABA control stomatal behavior. Cytokinin, gibberellin, and jasmonate sensors are also being refined, allowing researchers to track hormone dynamics across tissues. Although ethylene is gaseous and harder to visualize directly, innovative imaging approaches have enabled indirect visualization of ethylene signaling responses in live cells.
Advanced microscopy techniques are equally important. Confocal laser scanning microscopy provides high spatial resolution and allows optical sectioning of tissues. Spinning-disk confocal microscopy enables faster imaging with reduced photodamage, making it suitable for long-term live imaging. Light-sheet microscopy offers rapid 3D imaging with minimal stress to the sample, allowing hormone transport to be tracked across entire organs. Together, these tools allow scientists to watch hormone molecules and transporters move through cells, tissues, and organs as living processes unfold.
What Live-Cell Imaging Has Revealed About Hormone Transport
Live-cell imaging has overturned many traditional assumptions about hormone transport. One of the most striking discoveries is the extreme dynamism of hormone transporters. Auxin transporters such as PIN proteins constantly cycle between the plasma membrane and internal compartments. Their polarity determines the direction of auxin flow, but this polarity is not fixed. It can change rapidly in response to light, gravity, mechanical stress, or hormone feedback. During gravitropism, for example, live imaging shows how PIN proteins relocate within minutes, redirecting auxin flow to one side of the root or shoot. This asymmetric auxin distribution drives differential cell elongation and bending. Without live imaging, the speed and coordination of these events would be impossible to appreciate. Live imaging has also revealed how hormones move through tissues in waves rather than smooth gradients. ABA accumulation during drought can spread from vascular tissues outward, creating spatial patterns that fine-tune stomatal closure. Jasmonate signaling following wounding spreads as a coordinated signal through vascular networks, activating defense genes far from the injury site. In roots, live-cell imaging has shown how hormone transport interacts with root architecture. Auxin fluxes shape lateral root initiation sites, while cytokinins counterbalance auxin to regulate root branching. These interactions are highly dynamic and depend on environmental conditions such as nutrient availability and soil moisture. Importantly, live-cell imaging has demonstrated that hormone transport is tightly linked to cellular metabolism, cytoskeleton organization, and membrane trafficking. Hormone movement is not an isolated process but part of an integrated cellular system responding to internal and external signals.
Implications for Stress Biology, Crop Improvement, and the Future
The ability to visualize hormone transport in living plants has profound implications for stress physiology. Under drought, heat, salinity, or pathogen attack, plants must rapidly redirect hormonal signals to survive. Live-cell imaging allows researchers to see how these signals change in real time, revealing early stress responses long before visible symptoms appear. This knowledge is invaluable for crop improvement. Stress-tolerant varieties often differ not in how much hormone they produce, but in how efficiently hormones are transported and perceived. Faster ABA redistribution, more precise auxin gradients, or better coordination between hormone pathways can significantly enhance resilience. Live-cell imaging also supports modern breeding and biotechnology. When combined with CRISPR or prime editing, researchers can directly observe how genetic modifications alter hormone transport dynamics. This accelerates functional validation and helps design crops with optimized signaling networks rather than single-gene traits. Looking ahead, live-cell hormone imaging is likely to integrate with nanobiosensors, artificial intelligence, and digital plant models. Wearable sensors may one day report hormone dynamics in the field, while AI algorithms interpret these signals to guide precision agriculture. The vision is a future where plants themselves communicate their physiological state in real time.
Conclusion
Live-cell imaging of hormone transport has opened a window into the living plant that was unimaginable just a few decades ago. By allowing scientists to watch hormones move through cells and tissues in real time, this technology has transformed our understanding of plant development, stress responses, and adaptation. Hormones are no longer abstract signals inferred from gene expression or chemical assays. They are dynamic messengers whose journeys can now be seen, measured, and understood in their full complexity. As climate challenges intensify, this knowledge will be essential for designing crops that respond faster, smarter, and more efficiently to environmental stress. Live-cell imaging reminds us that plant physiology is not static, it is a constantly shifting landscape of signals in motion, shaping life one molecule at a time.