Stomata are tiny pores on the leaf surface, but their role in plant physiology is enormous. They regulate the exchange of gases allowing CO₂ to enter for photosynthesis while controlling water vapor loss. Yet behind each stomatal pore lies a pair of specialized cells known as guard cells, which function like intelligent hydraulic valves. These cells sense environmental signals such as light, drought, humidity, CO₂ levels, temperature and adjust their turgor pressure accordingly to open or close the pore.
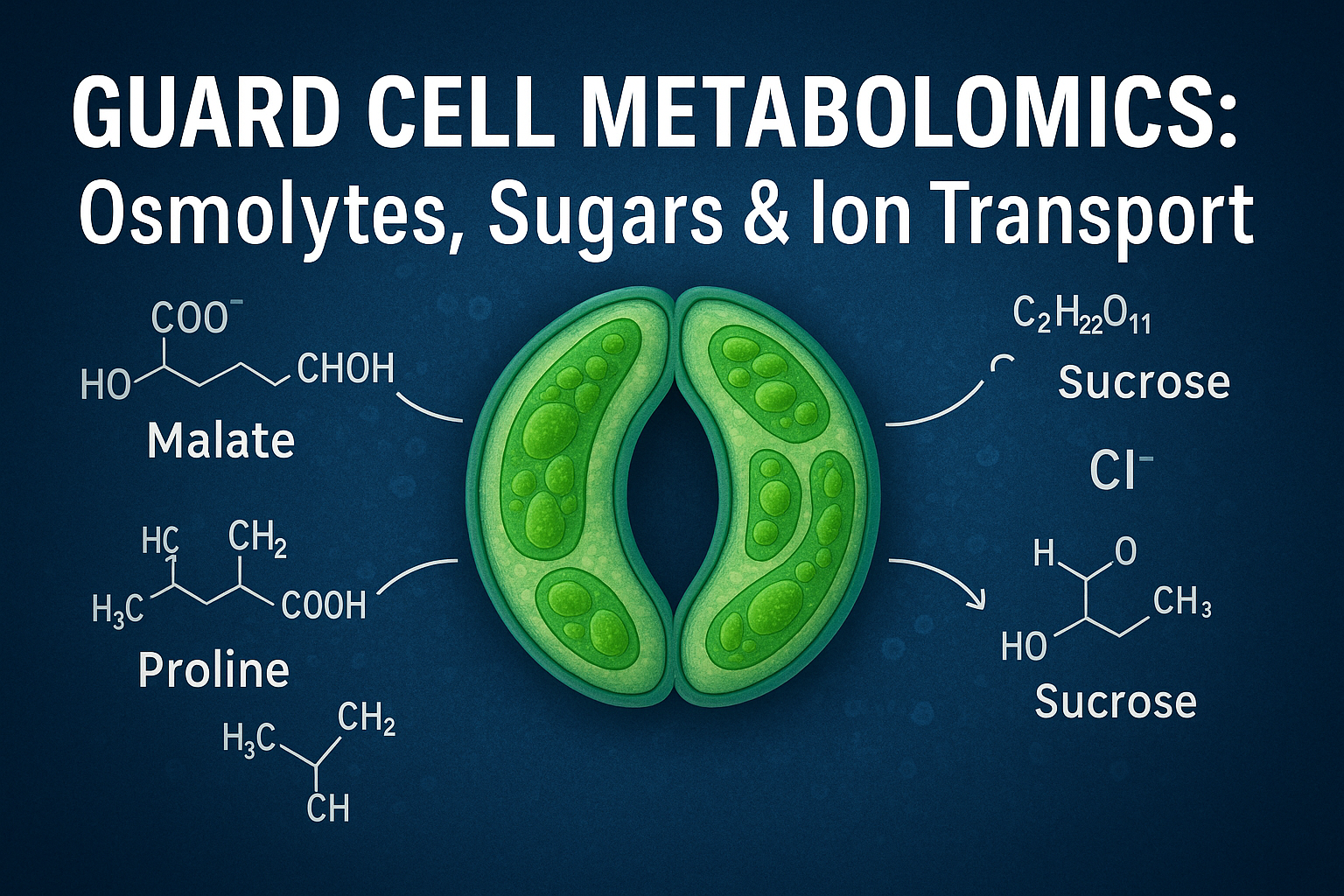
For decades, scientists viewed guard cell behaviour primarily as a result of ion movement, especially potassium. But today, thanks to metabolomics, it is clear that guard cells operate with far more complexity. They accumulate sugars, organic acids, amino acids, and a wide variety of osmolytes that help fine-tune stomatal movements. Guard cell metabolomics is revealing a dynamic biochemical landscape where ions, metabolites, and signaling pathways work together to determine whether stomata open wide for photosynthesis or close tightly to conserve water.
Understanding guard cell metabolism has become central to plant physiology, drought tolerance research, and efforts to engineer crops with improved water-use efficiency. This article explores how osmolytes, sugars, and ion transport integrate to control guard cell function and how these insights may transform the future of climate-resilient agriculture.
Guard Cell Osmolytes
At the heart of stomatal motion is turgor pressure the internal pressure that causes guard cells to swell or shrink. This pressure is regulated by osmolytes: molecules that help cells adjust their internal osmotic potential. Traditionally, potassium ions (K⁺) were considered the primary osmolytes involved in stomatal opening. When guard cells accumulate K⁺, water enters by osmosis, swelling the cells and opening the pore. When K⁺ leaves, water follows, the cells shrink, and the pore closes.
However, metabolomics has uncovered a more nuanced story. Guard cells accumulate a variety of organic osmolytes such as malate, fumarate, succinate, sucrose, and certain amino acids. These metabolites help maintain osmotic balance while providing metabolic flexibility. For example, in high light, guard cells generate malate as a counter-ion for K⁺, ensuring electrical neutrality while contributing to the buildup of osmotic potential. Under conditions where ionic uptake is limited, guard cells rely more heavily on sugar-based osmolytes.
Sucrose plays a particularly important role. As photosynthesis begins in the surrounding leaf tissue, sucrose levels rise. Some of this sucrose moves into guard cells and contributes to turgor. The presence of sucrose as an osmotic agent reduces the need for large K⁺ influxes. During the afternoon, sucrose accumulation can replace a significant portion of the osmotic load that was initially carried by K⁺, revealing a time-dependent shift in osmolyte composition.
Organic acids also participate in stomatal closure. When guard cells break down malate or convert sugars through metabolic pathways, the resulting decrease in osmolyte concentration promotes water efflux and pore closure. This dynamic interplay between synthesis, storage, and degradation of osmolytes allows guard cells to respond quickly and precisely to fluctuating environmental conditions.
Sugars as Metabolic Signals
Sugars inside guard cells do far more than regulate turgor. They act as metabolic signals that influence stomatal behaviour, linking leaf carbohydrate status with gas-exchange regulation. This connection ensures that stomata adjust not only to environmental cues but also to the internal metabolic needs of the plant.
Sucrose, for example, signals carbon sufficiency. When leaves generate abundant sugars under high light, sucrose levels rise in both mesophyll and guard cells. Elevated sucrose can promote stomatal closure as a form of feedback regulation, ensuring that the plant does not waste water when CO₂ supply becomes non-limiting. This feedback helps maintain water-use efficiency and prevent excessive transpiration during periods of high photosynthetic activity.
Hexoses such as glucose and fructose also influence guard cell behaviour through metabolic sensors like hexokinase. Guard cells detect intracellular sugar levels, and this information shapes stomatal responses. For instance, glucose accumulation can inhibit stomatal opening, while low sugar levels often encourage wider stomatal aperture to maximize CO₂ uptake.
Metabolomic analyses have shown that guard cells possess an active glycolytic network, meaning they generate ATP and reducing power dynamically as they adjust their aperture. The presence of functional chloroplasts within guard cells adds another regulatory layer. These chloroplasts engage in light-driven processes that contribute to osmolyte synthesis and signaling. Although guard-cell chloroplasts do not drive the bulk of leaf photosynthesis, their metabolic activity supports stomatal opening by powering proton pumps and synthesizing organic osmolytes.
Sugars also interact with hormonal signals. Abscisic acid (ABA), the drought hormone, modulates sugar metabolism in guard cells and shifts the balance away from osmolyte accumulation, promoting closure. Meanwhile, high sugar availability can reduce ABA sensitivity, making stomata more resistant to drought-induced closure. These interactions underline the importance of sugar metabolism as part of a complex, integrative network governing stomatal dynamics.
Ion Transport
Ion transport remains central to guard cell physiology, even as metabolomics reveals new biochemical layers. Stomatal opening begins with activation of proton pumps in the plasma membrane specifically, H⁺-ATPases that extrude protons out of the cell. This creates a negative membrane potential inside the guard cell, triggering inward movement of K⁺ through voltage-gated K⁺ channels. As K⁺ enters, guard cells accumulate counter-ions like chloride (Cl⁻) or organic acids such as malate²⁻ to maintain charge balance.
Water follows these ions through aquaporins, rapidly increasing cell volume and opening the pore. The speed at which stomata open is largely determined by the kinetics of ion channel activity and aquaporin regulation. Under bright light, many of these channels are activated simultaneously, allowing stomatal opening within minutes.
Stomatal closure involves a reversal of these processes. ABA signaling triggers Ca²⁺ waves within guard cells, activating anion channels that release Cl⁻ and malate. As negative ions exit, the membrane depolarizes, causing outward movement of K⁺ through K⁺ efflux channels. Water exits through aquaporins, reducing turgor pressure and closing the pore.
What metabolomics adds to this picture is a deeper understanding of how ions and osmolytes interact. For instance, when malate is abundant, guard cells rely less on Cl⁻. When sucrose replaces K⁺ as an osmolyte, ion channel activity shifts accordingly. Ion fluxes and metabolic pathways work together, creating a tightly integrated system where biochemical changes influence electrophysiological behaviour.
This integration becomes especially important under stress. During drought, ABA modifies ion channel sensitivity, accelerates Ca²⁺ signaling, and alters metabolite flows. Under high CO₂, guard cells reduce K⁺ uptake and shift metabolism to favour closure. Heat stress produces unique profiles where both ion fluxes and osmolyte synthesis adjust simultaneously to maintain water balance.
Thus, guard cell physiology is not controlled by ions alone, but by a dynamic partnership between ion transport, metabolism, and environmental signaling.
Implications for Climate-Resilient Agriculture
Understanding guard cell metabolomics has enormous practical significance. Stomata lie at the intersection of water use, CO₂ uptake, and stress tolerance. Crops with faster stomatal responses, more efficient osmolyte management, or improved ion channel regulation could thrive under variable climates.
Breeders and molecular biologists are already exploring multiple strategies: modifying proton pump activity to speed stomatal opening in low CO₂; altering ion-channel genes to improve drought-induced closure; engineering metabolic pathways so guard cells accumulate osmolytes more efficiently; adjusting sugar sensing to fine-tune water-use efficiency.
Recent research shows that reducing stomatal density can improve drought resilience, while maintaining fast guard-cell kinetics ensures that photosynthesis does not suffer under fluctuating light. Metabolomics provides the detailed biochemical map needed to guide these approaches.
As climate patterns shift, crops will need stomata that are responsive, conservative with water, and efficient in CO₂ uptake. Guard cell metabolomics—by revealing the biochemical dialogue inside each stomatal valve offers a roadmap to engineering plants that can survive and perform under future environmental extremes.