In the last decade, CRISPR revolutionized plant biology by giving scientists a simple, powerful way to edit DNA. But as our ambitions for crop improvement have grown from developing drought-tolerant varieties to reconfiguring metabolic pathways scientists have needed tools that are even more precise, flexible, and predictable than traditional CRISPR–Cas9 systems. This has led to one of the most significant innovations in gene editing: CRISPR Prime Editing.
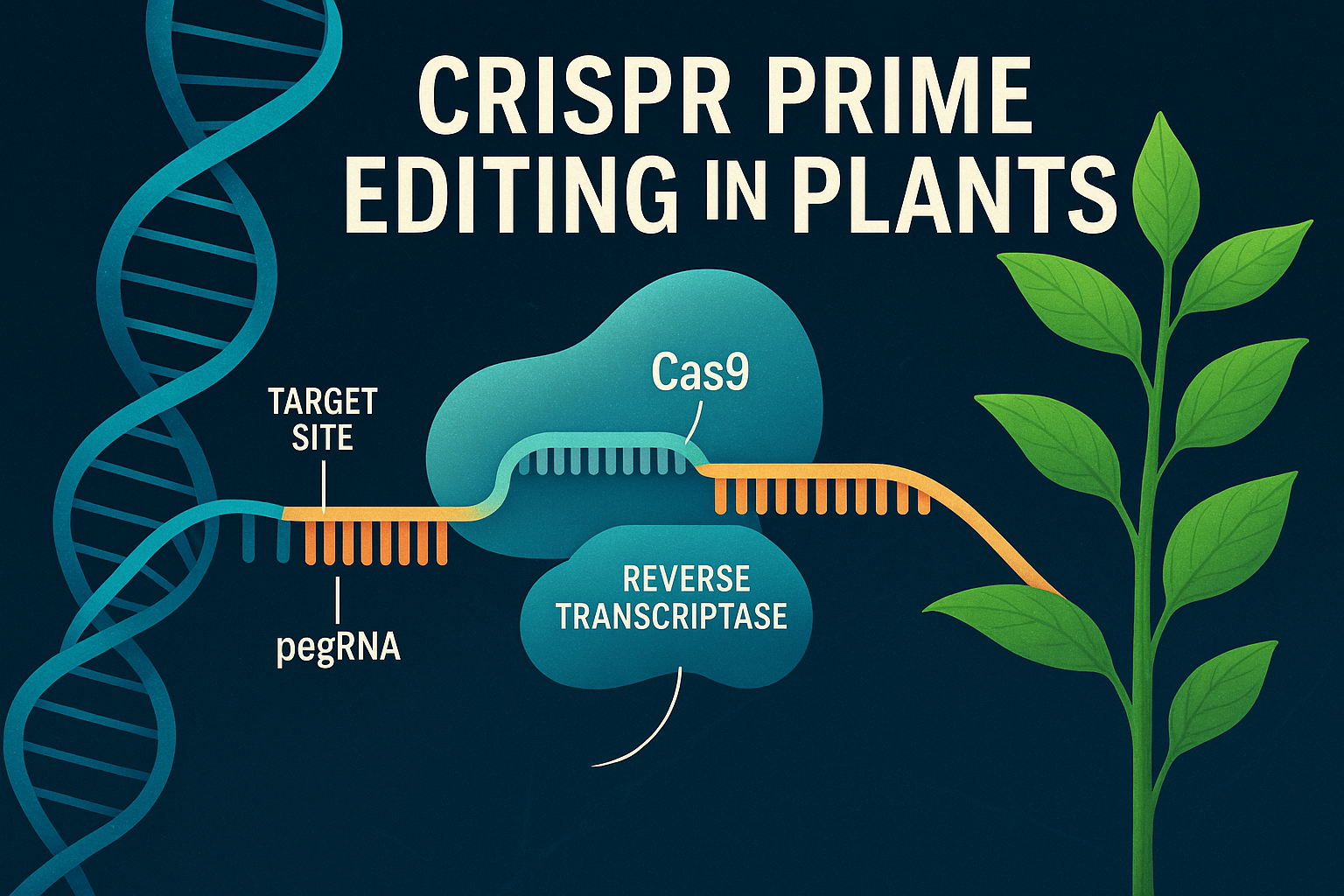
Prime editing is often described as a “genetic word processor.” Instead of cutting DNA in both strands like CRISPR–Cas9, prime editing rewrites DNA sequences using a reverse transcriptase fused to Cas9. This system allows plant scientists to create targeted insertions, deletions, and base substitutions without relying on error-prone repair pathways. Prime editing is cleaner, more precise, and capable of correcting or introducing nearly any type of genetic variation.
For agriculture, this opens extraordinary possibilities: designing crops with enhanced climate resilience, improving nutrient efficiency, eliminating susceptibility genes, fine-tuning metabolic pathways, and even resurrecting lost alleles from ancestral varieties. Prime editing represents the next generation of genome engineering a tool that closes the gap between what we can imagine and what we can genetically create.
What Is CRISPR Prime Editing?
Traditional CRISPR–Cas9 cuts DNA at a precise location, causing double-strand breaks (DSBs). While powerful, this mechanism relies on cellular repair systems that often introduce errors or unpredictable edits. Prime editing avoids DSBs entirely. Instead, it uses a nickase version of Cas9 (Cas9 H840A) fused to an engineered reverse transcriptase. This fusion protein is guided by a special RNA molecule called a prime editing guide RNA, or pegRNA, which carries both targeting information and the new genetic sequence to be written into the genome.
When the prime editor reaches its target site, it makes a single-strand nick and uses the pegRNA as a template to synthesize the desired edit directly onto the DNA. The cell then incorporates this newly synthesized sequence into the genome with minimal errors.
Prime editing introduces almost any type of substitution, insertion, or deletion. It reduces off-target mutations. It avoids the stress and genomic instability associated with double-strand breaks. It expands editing to genomic regions that were previously difficult to target. It enables subtle, precise modifications such as correcting a single base or altering protein function with surgical precision. For plant biotechnology, this means researchers now have a much more reliable tool for engineering crops that can withstand the complex challenges of a changing climate.
Applications in Plant Science
The potential uses of prime editing in plants span almost every aspect of physiology, development, and metabolism. One of the most exciting applications is the development of climate-resilient crops. Prime editing can target genes involved in drought tolerance, heat shock responses, stomatal development, osmotic regulation, or ROS detoxification. Unlike traditional breeding, which can take decades, prime editing introduces precise alleles directly into elite cultivars, accelerating the development of stress-resilient varieties.
Prime editing is also opening new possibilities for improving nutrient-use efficiency, especially nitrogen and phosphorus uptake. By fine-tuning regulatory regions of transporter genes, scientists can optimize nutrient absorption without increasing fertilizer input, reducing environmental impact. Another major application is disease resistance. Many plant pathogens exploit susceptibility (S) genes within the host. Prime editing can deactivate or modify these genes without introducing foreign DNA creating durable resistance that is indistinguishable from natural mutations. Similarly, resistance genes (R genes) can be strengthened or rewired for more effective immune responses.
Prime editing is also being used to modify photosynthesis-related genes, potentially enhancing Rubisco efficiency, chloroplast development, or photoprotective pathways. Small edits in promoter regions or catalytic sites can significantly influence how plants respond to light, CO₂ availability, or temperature.
In metabolic engineering, prime editing allows researchers to redesign pathways that control pigment production, flavor compounds, nutritional content, and secondary metabolites. For example, precise editing of enzymes in carotenoid or flavonoid pathways can elevate antioxidant content, improve color, or increase health benefits of fruits and vegetables. What makes all these applications so transformative is that prime editing can create non-transgenic modifications. Because it does not introduce foreign DNA, prime-edited plants may face fewer regulatory barriers and may be more readily accepted by consumers.
Technical Challenges and Breakthroughs
While prime editing is revolutionary, its early application in plants revealed challenges. Plant cells, with their rigid walls, complex chromatin structures, and unique DNA repair pathways, are not always as amenable to editing as mammalian cells. Initial efficiency rates were low, prompting intensive research to optimize the system.
One major advance has been the development of improved pegRNA designs. Researchers found that stabilizing the pegRNA, optimizing primer-binding sites, and adjusting extension lengths significantly enhances editing efficiency. Modified pegRNAs with protective structures, such as pseudoknots or MS2 loops, have further increased stability. Another breakthrough came from optimizing promoters that drive expression of Cas9 nickase and reverse transcriptase. Tissue-specific or strong viral promoters have improved editing in certain crops. Additionally, co-expression of specific DNA repair factors helps the cell integrate the edited strand more efficiently.
Recent studies have also explored temperature optimization, as some prime editing components perform better under warmer conditions useful for tropical crops. Researchers have begun combining prime editing with other CRISPR tools. For instance, PE3 systems include an additional guide RNA to nick the non-edited DNA strand, increasing incorporation efficiency. Base editors and prime editors can also be used together to create highly customized editing strategies. As these technical enhancements accumulate, prime editing is becoming increasingly reliable for a wide range of species, including rice, wheat, maize, tomato, potato, and fruit trees.
The Future of Prime Editing in Agriculture
Prime editing represents a major shift in how we think about plant improvement. Instead of crossing and selecting for years, or relying on random mutagenesis, prime editing allows scientists to introduce exact genetic edits that are predictable, stable, and physiologically meaningful. This opens the door to an agricultural future where crops are tailor-made to withstand extreme climates, flourish with fewer inputs, and contribute to sustainable food systems.
In the coming years, we can expect to see prime editing applied to improving carbon assimilation efficiency; tuning stomatal sensitivity and water-use efficiency; engineering resilience to combined heat + drought stresses; restoring lost alleles from wild ancestors; enhancing nutrient content for human health; producing crops suited to low-input, regenerative farming systems.
Advances in delivery technologies such as nanoparticle-mediated delivery, viral vectors, and DNA-free editing will make prime editing even more accessible. Eventually, prime editing platforms may integrate with machine learning models that predict optimal edits or simulate trait outcomes before experiments begin. Prime editing will not replace traditional breeding, but it will complement and accelerate it. By merging precision biotechnology with physiological insight, prime editing provides a powerful toolkit for designing crops that meet the demands of a warming and unpredictable world.
Conclusion
CRISPR Prime Editing marks the beginning of a new era in plant biology one where genetic changes can be made accurately, predictably, and without genomic scars. Its ability to rewrite DNA with surgical precision gives plant scientists a level of control that was unimaginable a decade ago. As the technology continues to evolve, its impact on crop improvement, stress resilience, and sustainable agriculture will only grow.
Prime editing is not just a scientific tool; it is a bridge between our understanding of plant physiology and our vision for the future of food security. By enabling targeted, minimal, and elegant genetic edits, prime editing empowers us to design crops that can thrive amid climate challenges and nourish a growing world.